If you have been living with psoriasis for a while, you may have already tried creams, tablets, or light therapy. For many people, these treatments help to manage flare-ups and keep symptoms under control. But for others, the results are disappointing, and the condition keeps coming back or getting worse despite trying multiple options.
If that sounds familiar, you might have wondered whether a psoriasis clinical trial could be an option worth exploring. This post explains exactly what is involved, who can take part, and what happens at each stage, so you can decide whether it is right for you.
What Is Psoriasis?
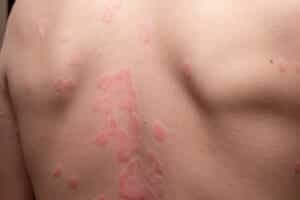
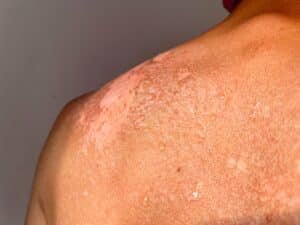
Psoriasis is a long-term skin condition that causes the skin to produce new cells far more quickly than usual. Normally, skin cells take around three to four weeks to move through the layers of skin and shed naturally. In people with psoriasis, that process can happen in as little as three to seven days. Because the cells are moving so fast, they build up on the surface before they are fully formed, creating the dry, flaky patches that are the main sign of the condition.
The most common type is plaque psoriasis, which causes raised, scaly patches on areas such as the elbows, knees, scalp, and lower back. These patches can be itchy or sore, and in more severe cases the skin around joints can crack and bleed.
According to the NHS psoriasis overview, psoriasis affects around two per cent of people in the UK. It is caused by a problem with the immune system and is not contagious, meaning it cannot be passed from one person to another. Symptoms tend to come and go over time, with periods of flare-up followed by calmer stretches. There is currently no cure. Treatment focuses on managing symptoms, but what works for one person may not work for another, and many people find themselves cycling through options over the years.
Why Would Someone Join a Clinical Trial?
A clinical trial tests whether a new medication or treatment is safe and effective. Researchers need people with the relevant condition to participate so that they can gather the evidence needed to understand whether a new treatment works and what effects it has.
For people whose psoriasis has not responded well to existing treatments, a trial can offer access to newer therapies that are not yet available on the NHS. These are treatments that have already gone through earlier stages of testing and are now being assessed in a larger group of people. This means participating can put you at the forefront of research that may eventually change how psoriasis is treated across the UK.
Participants also receive close medical monitoring throughout the process, which means any changes to your skin condition are tracked carefully by a trained clinical team. You will be seen regularly, and any new developments will be noted and acted upon. Many people find this level of attention to their health beneficial in itself.
It is worth being clear about payments: trials run by 4MCS do not pay participants to take part. However, reasonable travel expenses are covered so that joining a trial does not leave you out of pocket.
Who Can Take Part?
Every clinical trial has a set of eligibility criteria. These are the conditions you need to meet before you can join. For a psoriasis trial, the criteria usually cover things like:
| Eligibility Factor | Typical Requirement |
| Age | Usually 18 or over |
| Diagnosis | Confirmed plaque psoriasis |
| Severity | Moderate to severe symptoms |
| Previous treatment | May need to have tried certain treatments already |
| Current medication | Some medications may exclude you temporarily |
| General health | No serious unrelated conditions in most cases |
These criteria exist to protect participants and to make sure the trial produces reliable results. They are set in advance by the researchers and approved by an independent ethics committee before the trial starts.
You will not know for certain whether you qualify until you have been through a screening visit. Not everyone who enquires will be accepted onto a trial, and that is completely normal. It does not reflect on you as a person, and it does not mean you cannot enquire again in the future if circumstances change.
What Happens at a Screening Visit?
Before you can join a trial, you will be invited to a screening visit. This is a medical appointment where the clinical team checks whether you meet the eligibility criteria for the specific study.
You can read a full breakdown of what to expect at a screening visit on the 4MCS website. In general, the visit will include:
- A review of your medical history and any previous treatments you have used
- A physical examination, including an assessment of your skin
- Blood tests or other routine health checks
- A thorough explanation of the trial, including what it involves, how long it lasts, and any known risks or potential side effects
- Time to ask questions and speak with the clinical team before making any decision
The screening visit is also when you will be given the participant information sheet for the trial. This is a document that sets out everything you need to know in plain language. You are encouraged to take it away, read it carefully, and discuss it with family or friends if that would help.
At no point are you committed to anything at the screening stage. You are free to decide not to take part after the visit, and this will not affect any other care you receive from the NHS.
What Does the Trial Itself Involve?
If you are accepted onto a psoriasis trial, the process will vary depending on the specific study. Some trials test an injection given every few weeks, while others may involve a daily tablet or cream. The duration and number of visits will also differ. However, most trials follow a similar overall pattern:
Baseline assessment — Before any treatment begins, the team will take detailed measurements of your skin condition. This might include scoring the severity of your plaques, taking photographs, or asking you to complete a questionnaire about how your skin is affecting your daily life. This gives a clear starting point to compare against later in the trial.
Treatment phase — You will receive the treatment being tested. In some trials, a portion of participants receive a placebo, which is a treatment that looks identical to the real thing but contains no active medicine. Whether the trial uses a placebo and whether you would know which you had received will always be explained clearly before you sign up. In many psoriasis trials, all participants receive an active treatment at some point during the study.
Regular check-up visits — Throughout the trial, you will attend the clinic at set intervals, which are agreed in advance. At each visit, the team will examine your skin, check your overall health, and record any changes or side effects. These visits are also an opportunity for you to raise any questions or concerns.
End of trial — Once the treatment period ends, the team will carry out a final assessment. You will receive a summary of how things went, and if the treatment has produced a positive result, the clinical team may be able to discuss what happens next.
Is It Safe?
This is the most common concern people raise, and it is a fair question. Clinical trials in the UK are regulated by the Medicines and Healthcare products Regulatory Agency (MHRA). This means no trial can take place without passing through independent ethical approval and meeting strict safety standards.
You can find out more about how clinical trials keep participants safe in the 4MCS information hub. Some of the key protections include:
- All trials are reviewed and approved by an independent ethics committee before they begin
- You have the right to withdraw from the trial at any time and for any reason, without it affecting your NHS care
- Any side effects or unexpected reactions must be recorded and reported, and the trial team will respond quickly
- The clinical team is medically trained and is available to you throughout the process
- Trials are monitored continuously, and can be paused or stopped if any safety concerns arise
No medical treatment, including those already widely available on the NHS, is entirely without risk. But clinical trials are among the most closely monitored medical activities that exist in the UK.
How Long Does a Psoriasis Trial Last?
Trial lengths vary depending on what is being tested. Some studies run for a few months, while others may last a year or more. The specific duration will always be explained clearly before you sign up, so you can plan around it and decide whether it fits with your life.
Most visits do not take the whole day, and many can be scheduled at a time that suits you. The 4MCS clinics are based in Swinton, Greater Manchester and Ilford, East London, both of which are well served by public transport. Reasonable travel expenses are reimbursed.
Common Concerns
“I am worried about being given a placebo.” This is understandable. The use of placebos is carefully controlled and always fully explained before you sign up. No participant is left without information about what they are receiving. In many psoriasis trials, all participants receive an active treatment, either throughout the study or during a particular phase of it.
“I do not want to stop my current medication.” Some trials do require a washout period, where you pause certain treatments for a set amount of time before the study begins. This is because some medications can interfere with the results. Whether this applies and how long any washout period would be will be discussed at your screening visit, giving you everything you need to make an informed decision.
“I am not sure I have the time.” Visit schedules are agreed in advance and designed to be as manageable as possible. The clinical team will always try to work around your circumstances, and visits are typically spaced out rather than frequent. If you have specific constraints around work or travel, it is worth raising these when you first get in touch.
“What if the treatment does not work for me?” Not every trial produces the hoped-for result for every participant. If the treatment does not work, your safety and wellbeing remain the priority throughout. You will not be left without support, and you can return to your usual care through your GP or dermatologist.
What Makes 4MCS Different?
4MCS is a UK clinical research organisation with a combined team experience of over 130 years in clinical trials and the pharmaceutical industry. The team runs MHRA-approved trials across a range of conditions, with dedicated clinic sites in Manchester and London.
The focus at 4MCS is on making the experience as straightforward and comfortable as possible for participants. The clinics are set up to feel welcoming rather than clinical, and the staff are experienced in working with people who are new to research. You will always have a named member of the team to speak to, and you will never be left unsure about what is happening next.
If you have questions about the process before you even make an initial enquiry, that is absolutely fine. The team is used to people wanting to understand the process thoroughly before committing to a screening visit. There is no pressure and no rush.
Ready to Find Out More?
If you have moderate to severe plaque psoriasis and want to find out whether you could be eligible, the best first step is to get in touch with the team at 4MCS. They can talk through your situation, explain which trials are currently open, and let you know whether a screening visit would be appropriate.
You can call on 03300 575 838 or email info@4mcs.co.uk.