Clinical research plays a vital role in developing new treatments and improving health and social care for everyone. Without people willing to take part, medical advances simply wouldn’t happen. If you’re thinking about joining a clinical trial, this guide will explain everything you need to know about taking part in clinical studies in the UK.
What is a clinical study?
 A clinical study involves research that tests new treatments, medicines, or medical devices on real people. These research studies help doctors understand what works, what doesn’t, and how to make healthcare better for patients everywhere.
A clinical study involves research that tests new treatments, medicines, or medical devices on real people. These research studies help doctors understand what works, what doesn’t, and how to make healthcare better for patients everywhere.
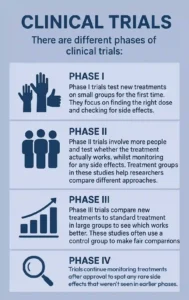
There are different phases of clinical trials:
Phase I trials test new treatments on small groups for the first time. They focus on finding the right dose and checking for side effects. These studies aim to understand how the body processes new treatments.
Phase II trials involve more people and test whether the treatment actually works, whilst monitoring for any side effects. Treatment groups in these studies help researchers compare different approaches.
Phase III trials compare new treatments to standard treatment in large groups to see which works better. These studies often use a control group to make fair comparisons.
Phase IV trials continue monitoring treatments after approval to spot any rare side effects that weren’t seen in earlier phases.
Who can take part?
Anyone can potentially become a participant in clinical research, whether you’re healthy or have a medical condition. Different research studies need different types of participants:
Healthy participants are people without the disease or condition being studied. You might help researchers understand how a new treatment affects healthy people, or provide comparison data for treatment groups.
Patient participants have the specific disease or condition being researched. You might get access to new treatments before they become standard treatment options.
Benefits of taking part in clinical trials
Taking part in clinical trials offers several advantages:
- You contribute to medical research that could help millions of people
- You might receive cutting-edge treatments before they become standard treatment
- You get extra medical attention and monitoring from expert healthcare teams
- You gain a better understanding of your health condition
What to consider before taking part
Clinical research isn’t right for everyone, and it’s important to understand both the benefits and risks. Each opportunity is different:
Time commitment: Some clinical trials require regular visits over weeks, months, or even years.
Possible side effects: New treatments may have unknown side effects, though researchers take every precaution to minimise risks. All known side effects will be explained clearly.
No guarantee: The new treatment might not work better than standard treatment, or might not work for you at all.
Control group possibility: Some studies use a control group that receives either a dummy treatment or standard treatment, so you might not receive the new active treatment.
How to find clinical trials
The best website to find clinical trials in the UK is the NHS Be Part of Research website. This National Institute for Health and Care Research (NIHR) service helps people find research studies across the country.
You can also:
- Ask your GP or specialist about relevant clinical trials
- Contact research centres directly
- Check with medical charities related to your condition
- Look at hospital websites, as many run their own clinical research
The process of joining a clinical trial
Here’s what typically happens when you join a clinical study:
- Initial contact: You’ll speak with the research team about the clinical trial and whether you might be suitable.
- Information session: Researchers explain the research study in detail, including what’s involved, potential side effects, and benefits.
- Screening: You’ll have tests to confirm you meet the study requirements.
- Application forms: You’ll complete application forms with your medical history and personal details.
- Informed consent: If you decide to take part, you’ll sign consent forms. Remember, you can withdraw from any clinical study at any time.
- Participation: You’ll follow the study protocol, which might include taking medication, attending appointments, or completing questionnaires.
- Follow-up: Many clinical trials continue monitoring participants even after the main research study ends.
Your rights as a participant
UK clinical trials are strictly regulated by the regulatory authority to protect participants. You have the right to:
- Receive clear, understandable information about the clinical study
- Take time to decide whether to take part in clinical research
- Ask questions at any stage about the research study
- Withdraw from clinical trials at any time without affecting your normal healthcare
- Access your study results if you want them
- Receive compensation if you’re harmed due to participation
All clinical studies must be approved by ethics committees and the regulatory authority before they can begin.
New regulations coming in 2026
The UK’s clinical trial regulations are being updated to make medical research more transparent and accessible. From April 2026, all clinical studies must be registered on public databases, and results must be published within 12 months of completion. This means it will be easier for people to find clinical trials and understand their outcomes.
How 4MCS can help
At 4MCS, we’re committed to advancing medical research through high-quality clinical trials. With over 130 years of combined experience in clinical studies and pharmaceutical research, we offer a comfortable, professional environment for participants in our research studies.
Our current clinical trials
We currently have several exciting clinical trials recruiting participants:
- Asthma clinical research: If you have asthma, you might be eligible for our asthma clinical study looking at new treatment approaches.
- Heart health studies: Our atrial fibrillation clinical trial is looking for participants with this heart rhythm condition who aren’t currently on standard treatment.
- Mental health research: We’re conducting depression clinical studies for people who haven’t found relief with standard treatment like medication or talking therapy.
- Pain management: Our lower back pain research study focuses on alternatives for people experiencing side effects from pain medications.
- Neurological conditions: We’re running clinical trials for people who suffer from migraines and people with myasthenia gravis, a rare muscle weakness condition.
- Women’s health research: Our clinical research includes studies on how hormonal changes affect women’s health and wellbeing.
- Skin condition studies: We have ongoing psoriasis clinical trials testing new approaches to this chronic skin condition.
Our comprehensive services
Beyond clinical trials, 4MCS offers a full range of clinical research services including clinical development and R&D support, specialised clinical site services, professional project management, pharmaceutical manufacturing expertise, medical writing and protocol development, central reading services, and laboratory services.
Getting involved with 4MCS clinical research
Our research facility in Ilford provides easy access for the over 10 million people living in the surrounding areas. We maintain a database of over 35,000 patients with various conditions, making it easier to match participants with suitable clinical studies.
If you’re interested in taking part in our clinical trials, our friendly, professionally trained staff will ensure you feel comfortable and well-informed throughout the entire process. We understand that deciding to become a participant is a big decision, and we’re here to support you every step of the way.
Taking the next step
Taking part in clinical trials is a meaningful way to contribute to medical research whilst potentially benefiting your own health and supporting public health. Whether you’re a healthy participant or have a medical condition, there are likely clinical studies that could use your help.
Remember, taking part in clinical research is always your choice, and you can change your mind at any time. The most important thing is that you feel comfortable and fully informed about what each research study involves.
If you’re ready to explore this opportunity, start by visiting the Be Part of Research website or speaking with your healthcare provider. You could be part of the next breakthrough in medical research.




